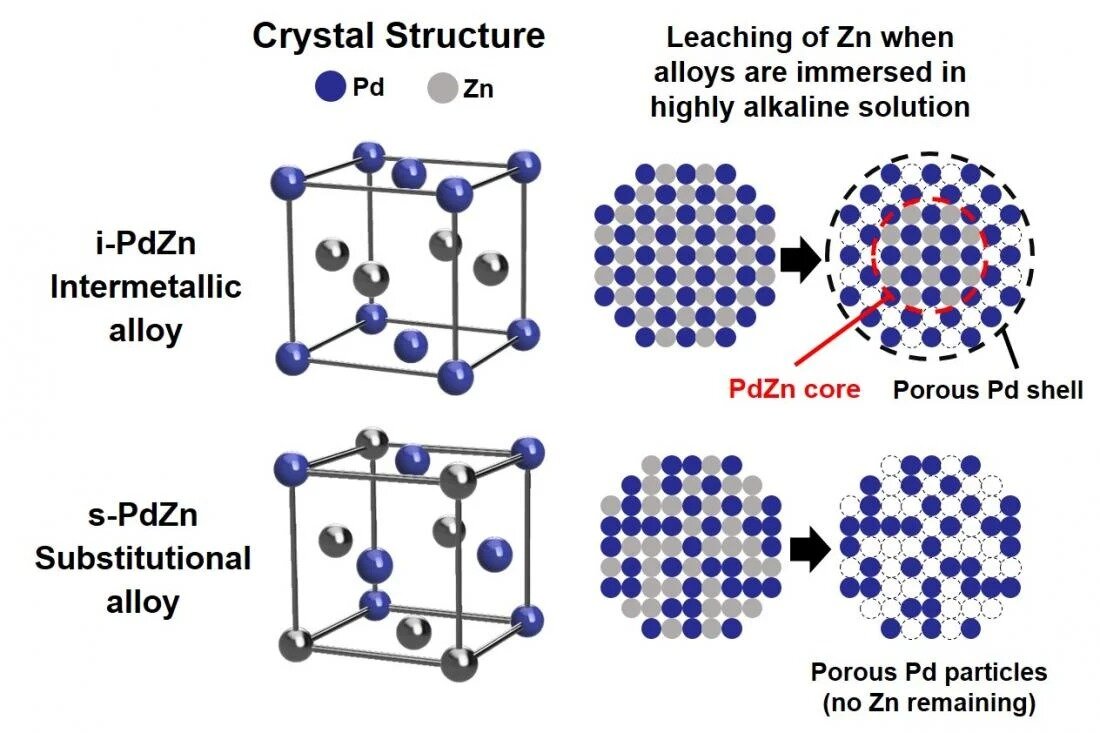
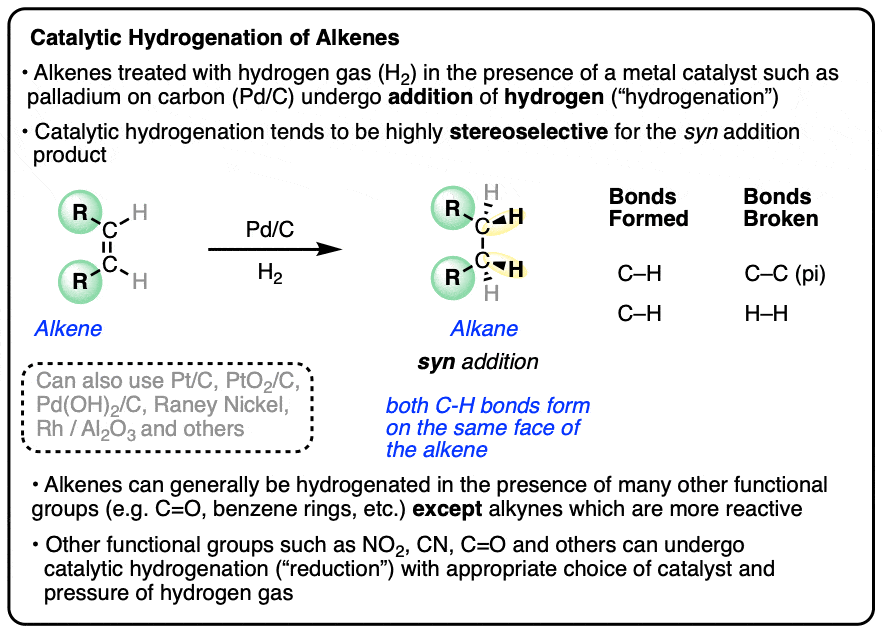
How palladium inhibits CO poisoning during electrocatalytic formic acid oxidation and carbon dioxide reduction | Nature Communications
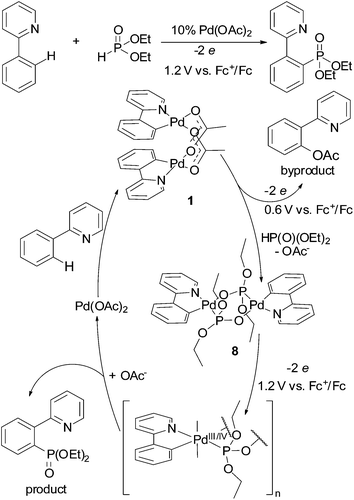
Redox trends in cyclometalated palladium( ii ) complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03786K

Redox trends in cyclometalated palladium( ii ) complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03786K

Tuning the Copper(II)/Copper(I) Redox Potential for More Robust Copper‐Catalyzed C–N Bond Forming Reactions - Cope - 2020 - European Journal of Inorganic Chemistry - Wiley Online Library

Tuning the Stability of Pd(IV) Intermediates Using a Redox Non-innocent Ligand Combined with an Organolanthanide Fragment | Journal of the American Chemical Society
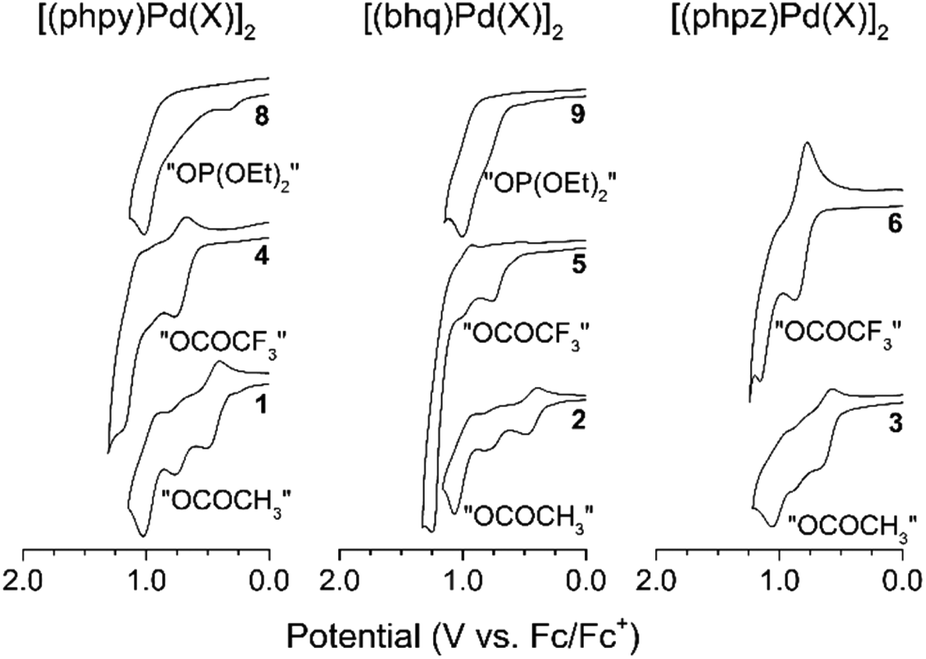
Redox trends in cyclometalated palladium( ii ) complexes - Dalton Transactions (RSC Publishing) DOI:10.1039/C6DT03786K

Can Donor Ligands Make Pd(OAc)2 a Stronger Oxidant? Access to Elusive Palladium(II) Reduction Potentials and Effects of Ancillary Ligands via Palladium(II)/Hydroquinone Redox Equilibria | Journal of the American Chemical Society

Hydrometallurgical recycling of palladium and platinum from exhausted diesel oxidation catalysts - ScienceDirect

Redox Properties of Zerovalent Palladium Complexes Containing α-Diimine and p-Quinone Ligands | Organometallics

Catalysts | Free Full-Text | Recent Advances in Platinum and Palladium Solvent Extraction from Real Leaching Solutions of Spent Catalysts

Aerobic Heterogeneous Palladium-Catalyzed Oxidative Allenic C−H Arylation: Benzoquinone as a Direct Redox Mediator between O2 and Pd | CCS Chemistry

Revealing the Influence of Diverse Secondary Metal Cations on Redox‐Active Palladium Complexes - Golwankar - 2022 - Chemistry – A European Journal - Wiley Online Library
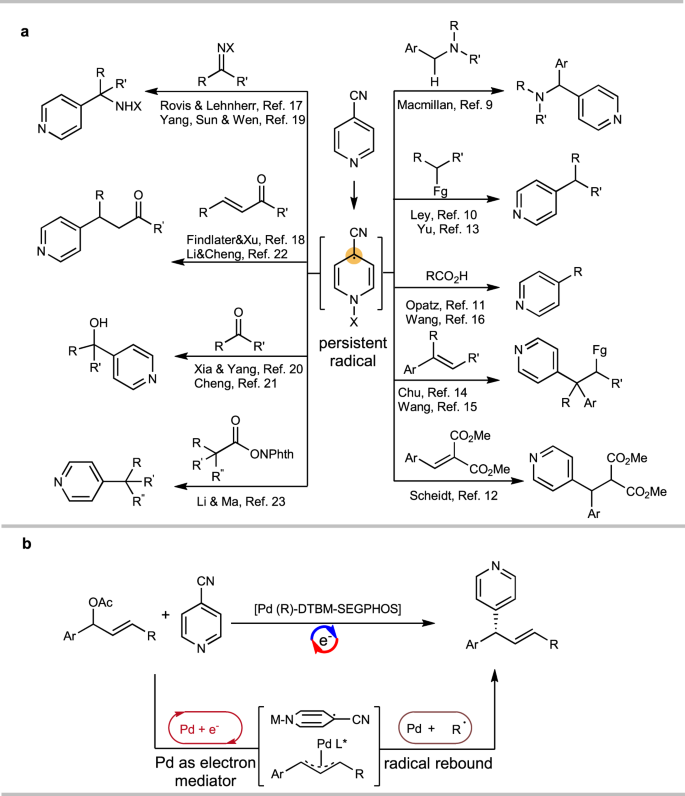
Palladium-catalyzed asymmetric allylic 4-pyridinylation via electroreductive substitution reaction | Nature Communications

A Distinctive Pattern for Substituent Effects on Transition Metal Centers: Enhanced Electron-Donating Capacity of Cationic Palladium Species | CCS Chemistry
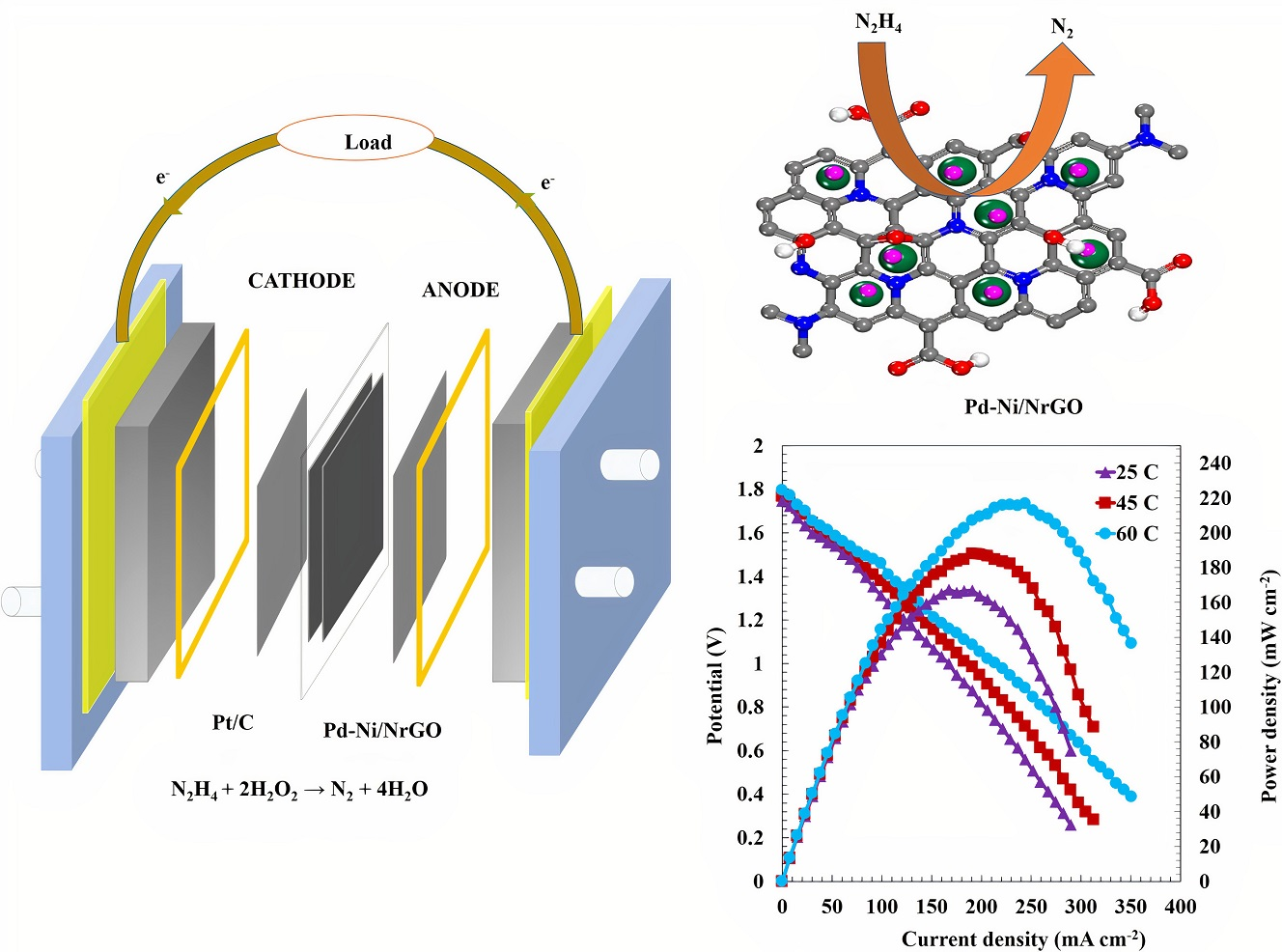
Catalysts | Free Full-Text | Palladium-Nickel Electrocatalysts on Nitrogen-Doped Reduced Graphene Oxide Nanosheets for Direct Hydrazine/Hydrogen Peroxide Fuel Cells

Can Donor Ligands Make Pd(OAc)2 a Stronger Oxidant? Access to Elusive Palladium(II) Reduction Potentials and Effects of Ancillary Ligands via Palladium(II)/Hydroquinone Redox Equilibria | Journal of the American Chemical Society
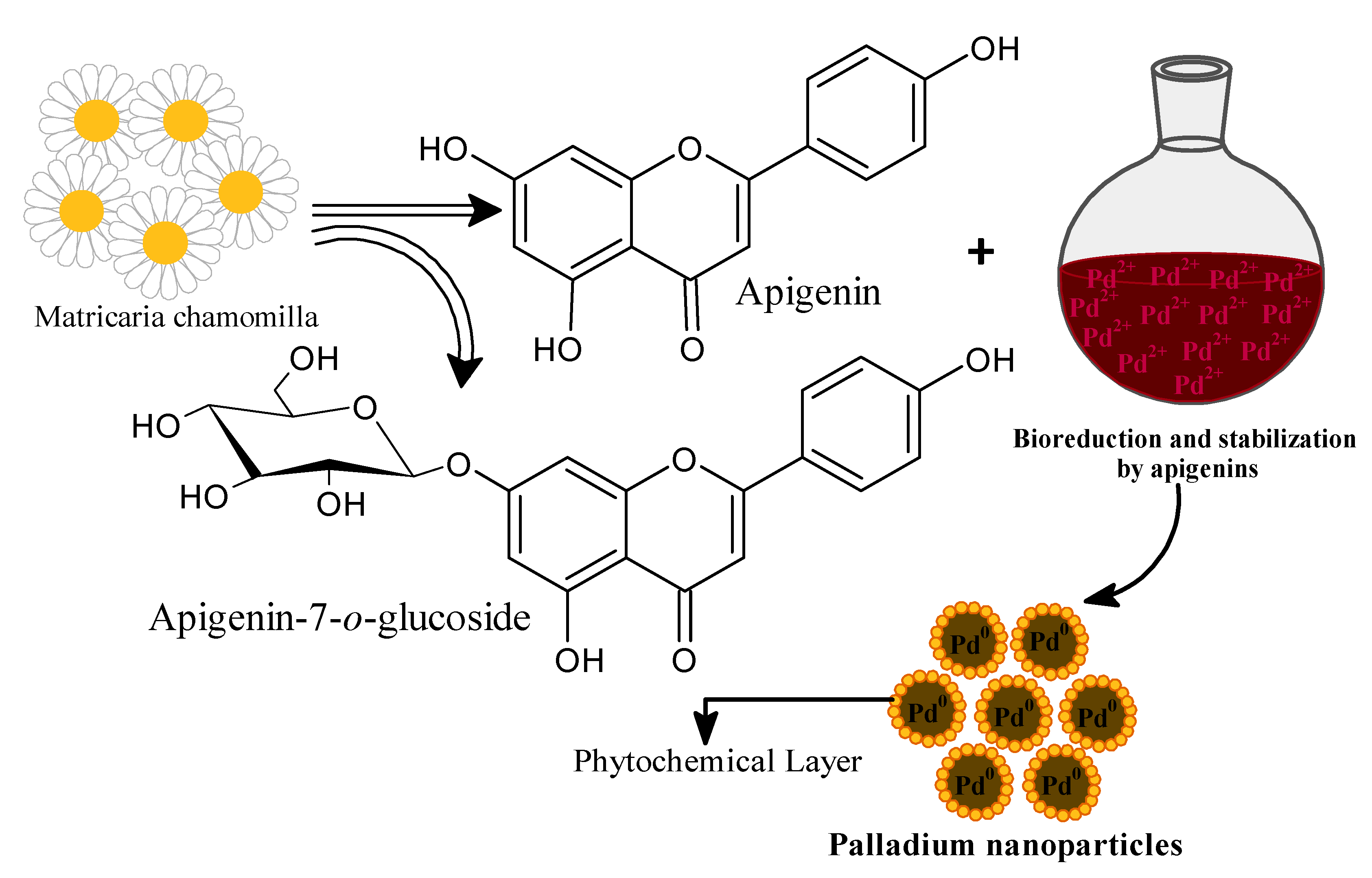
Toxics | Free Full-Text | Bioengineered Matricaria recutita Extract-Assisted Palladium Nanoparticles for the Congo Red Dye Degradation and Catalytic Reduction of 4-Nitrophenol to 4-Aminophenol

Can Donor Ligands Make Pd(OAc)2 a Stronger Oxidant? Access to Elusive Palladium(II) Reduction Potentials and Effects of Ancillary Ligands via Palladium(II)/Hydroquinone Redox Equilibria | Journal of the American Chemical Society
CV and redox potential for the Fe(III)/Fe(II) redox couple of different... | Download Scientific Diagram

Chemically activating MoS2 via spontaneous atomic palladium interfacial doping towards efficient hydrogen evolution | Nature Communications

Can Donor Ligands Make Pd(OAc)2 a Stronger Oxidant? Access to Elusive Palladium(II) Reduction Potentials and Effects of Ancillary Ligands via Palladium(II)/Hydroquinone Redox Equilibria | Journal of the American Chemical Society

In-situ oxidation of Palladium–Iridium nanoalloy anchored on Nitrogen-doped graphene as an efficient catalyst for methanol electrooxidation - ScienceDirect