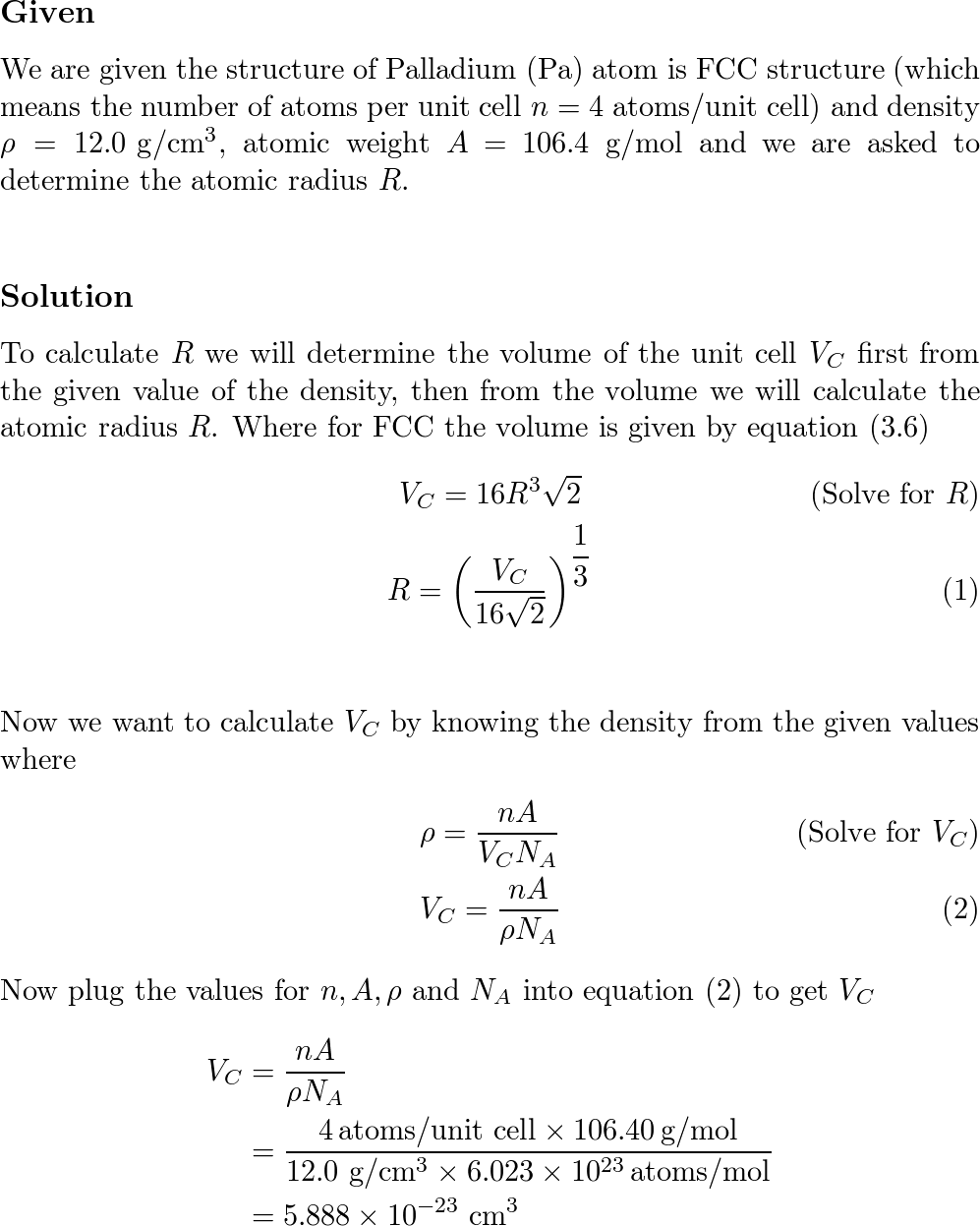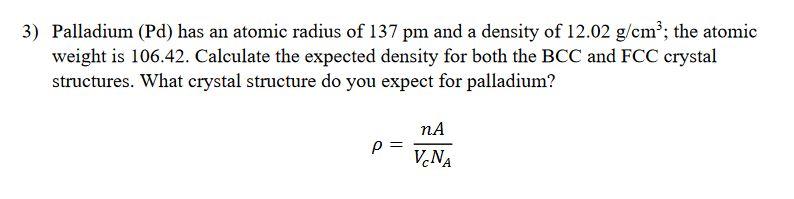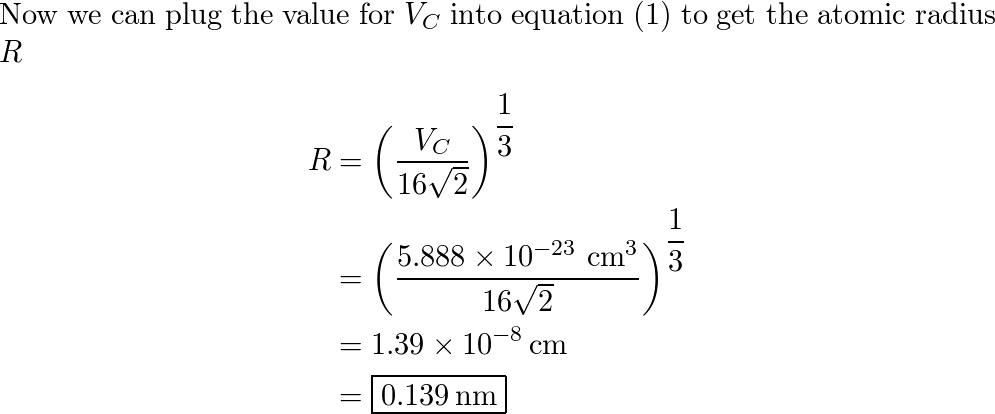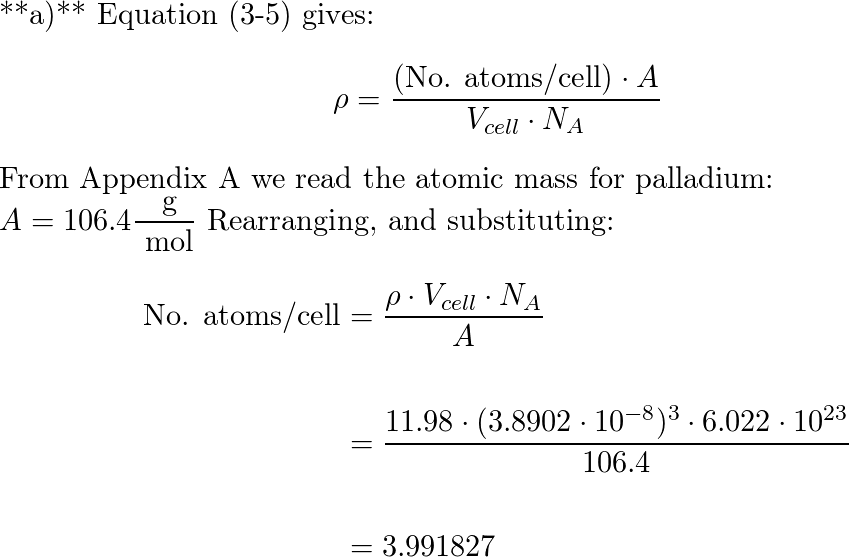Palladium Chloride Density: 4 G/Cm Gram Per Cubic Centimeter(G/Cm3) at Best Price in Mumbai | Indian Platinum Private Limited

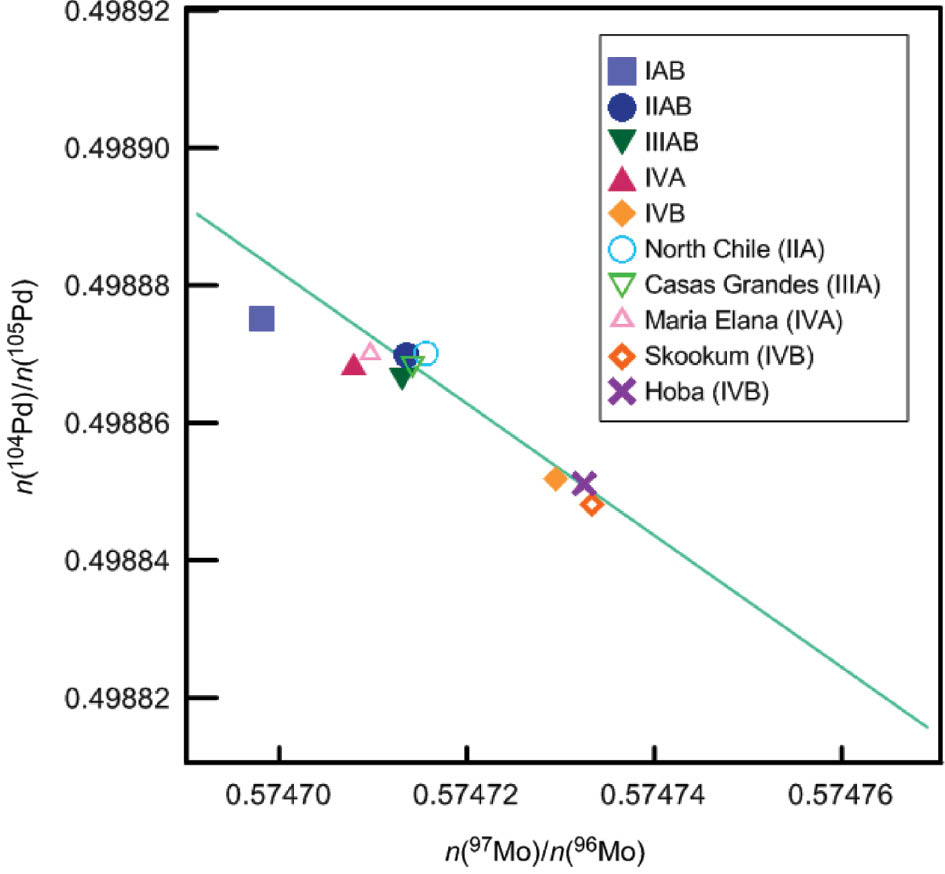
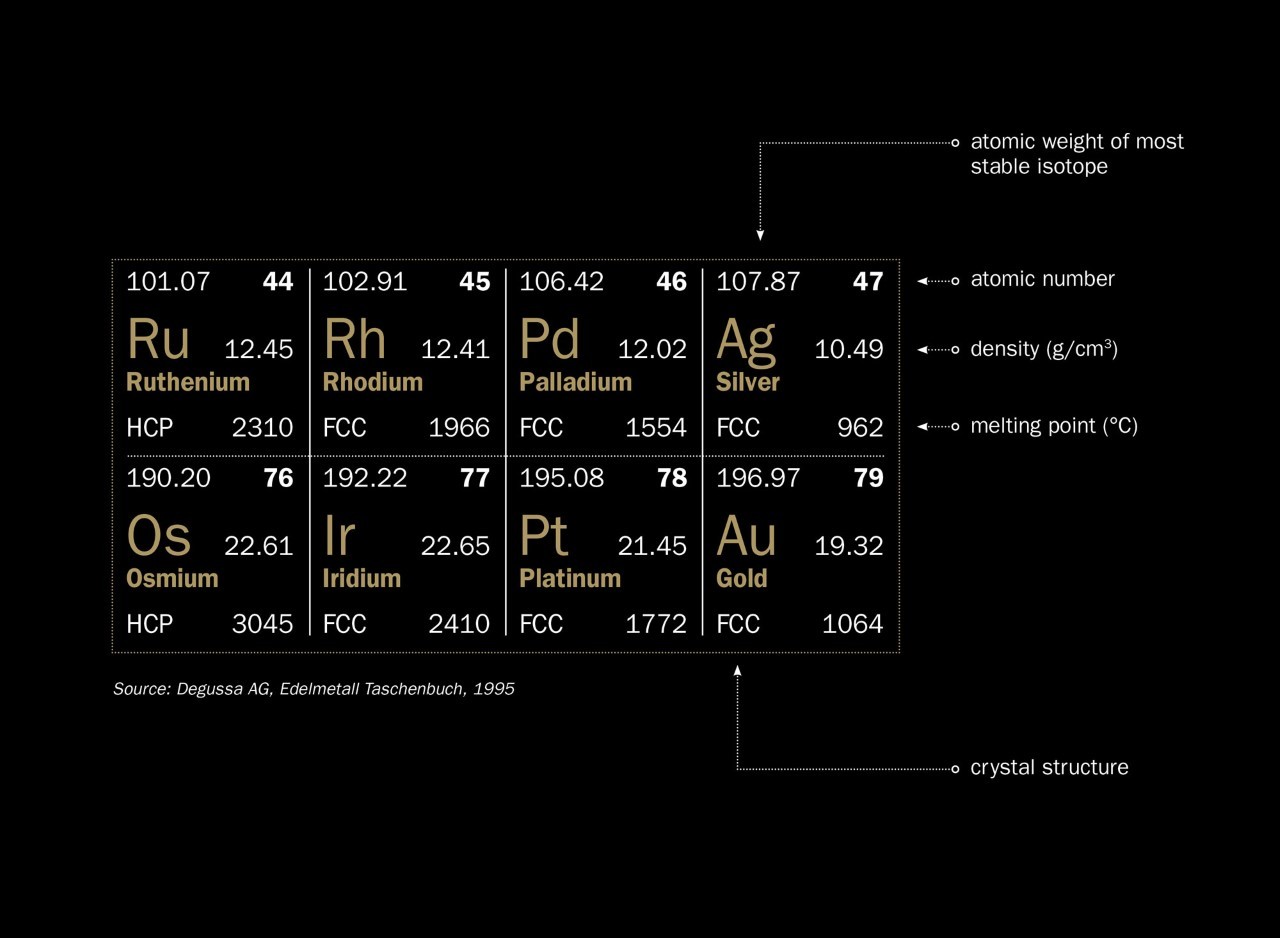
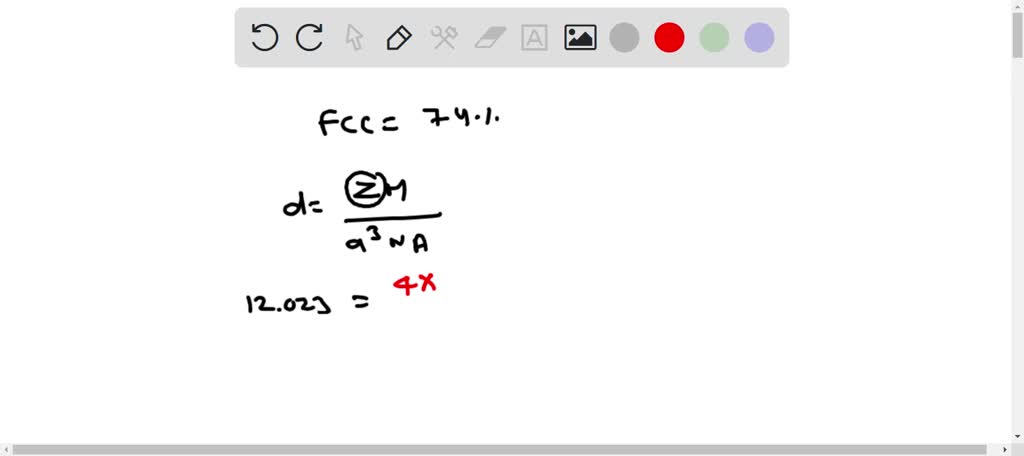
SOLVED: Calculate the radius of a palladium atom, given that Pd has an FCC crystal structure, a density of 12.0 g/cm3 , and an atomic weight of 106.4 g/mol.

Palladium and Pt atomic fractions and oxidation states calculated for... | Download Scientific Diagram

Palladium Alumina Catalyst Density: 12.02 G/cm3 Gram Per Cubic Centimeter(g/cm3) at Best Price in Mumbai | Indian Platinum Private Limited


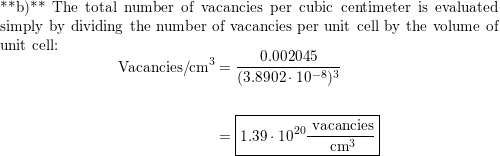
:max_bytes(150000):strip_icc()/palladium-56a129313df78cf77267f7e3.jpg)